Comparing 15 vendors in Clinical Trials Startups across 0 criteria.
The clinical trials market is a rapidly evolving landscape that is pivotal to the development of new therapeutic solutions. The market, projected to reach USD 101.63 billion by 2030 from USD 66.45 billion in 2025, showcases a robust compound annual growth rate (CAGR) of 8.9% over the forecast period. This rapid expansion can be attributed to several driving factors, including the burgeoning focus on precision and personalized medicine, increased research and development (R&D) investments, and significant technological advancements. These factors reflect the industry's commitment to innovation and efficiency in drug development processes.
Emerging trends such as in silico clinical trials are setting new benchmarks in the industry. These digital simulations can significantly cut research and development costs by over 50%, providing faster and more affordable solutions while enhancing the accuracy and safety of drug testing. Furthermore, the integration of artificial intelligence (AI) and machine learning in clinical trials enhances patient recruitment processes and trial design, offering predictive insights that improve trial outcomes. These technologies are crucial in addressing the rising complexities and costs associated with traditional clinical trials.
Despite these advancements, the market faces challenges such as cybersecurity risks and intellectual property concerns, which could impede its growth. Ensuring patient data protection and the ethical use of AI remains a priority as the industry navigates these hurdles. The market's dynamic nature is further influenced by regulatory requirements to increase clinical trial diversity, which urges companies to adopt more inclusive strategies.
Competition remains intense, driven by numerous global, regional, and niche Contract Research Organizations (CROs) vying for market share. Major players are continuously innovating through mergers and acquisitions to enhance their service offerings and expand their global footprint. The strategies adopted by these companies, such as forming strategic partnerships and developing comprehensive service portfolios, are aimed at maintaining competitive advantage and ensuring sustainable growth.
Key Developments
On October 2024, ProPharma has appointed Brian Tuttle as its Chief Financial Officer to lead financial strategy and support the company's growth. With over 20 years of experience in the life sciences sector, Tuttle previously served as CFO at LabConnect and Worldwide Clinical Trials, and spent two decades at PPD in various finance roles. He has a proven track record in financial transformation, systems integration, and strategic development.
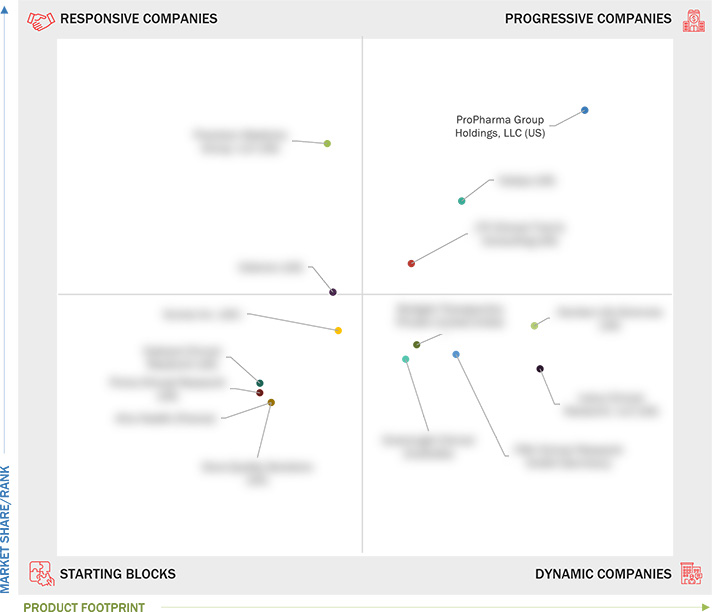
Market Leadership Quadrant
1.1 Study Objectives
1.2 Market Definition
1.3 Study Scope
1.3.1 Markets Covered and Regional Scope
1.3.2 Inclusions and Exclusions
1.3.3 Years Considered
1.4 Currency Considered
1.5 Unit Considered
1.6 Limitations
1.7 Stakeholders
2.1 Introduction
2.2 Market Dynamics
2.2.1 Drivers
2.2.1.1 Increasing focus on patient-centric trials (DCTs)
2.2.1.2 Increasing clinical trials for precision/personalized medicines supporting growth in clinical trial outsourcing
2.2.1.3 Rising R&D investments and patent cliff pressure driving growth in outsourcing clinical trial activities
2.2.1.4 Service flexibility offered by CROs for clinical development(FSP, FSO, Hybrid)
2.2.1.5 Technological integrations
2.2.2 Opportunities
2.2.2.1 Increased focus on pediatric clinical trials
2.2.2.2 Increasing regulatory requirements for increasing diversity in clinical trials
2.2.2.3 Growing role of real-world evidence in drug approvals
2.2.2.4 Expanding drug modalities (such as CGT, Tissue Engineering, Bispecific Abs) in clinical trials
2.2.3 Challenges
2.2.3.1 Cybersecurity or intellectual property concerns
2.2.3.2 Challenge of patient retention
2.2.3.3 Growing market competition
2.3 Trends/Disruptions Impacting Customer Business
2.4 Value Chain Analysis
2.5 Ecosystem Analysis
2.6 Investment and Funding Scenario
2.7 Technology Analysis
2.7.1 Key Technologies
2.7.2 Complementary Technologies
2.7.3 Adjacent Technologies
2.8 Patent Analysis
2.9 Trade Analysis
2.10 Porter’s Five Forces Analysis
2.10.1 Threat of New Entrants
2.10.2 Threat of Substitutes
2.10.3 Bargaining Power of Suppliers
2.10.4 Bargaining Power of Buyers
2.10.5 Intensity of Competitive Rivalry
3.1 Introduction
3.2 Key Player Strategies/Right to Win
3.3 Revenue Analysis
3.4 Market Share Analysis
3.5 Company Valuation and Financial Metrics
3.6 Brand/Product Comparison
3.7 Company Evaluation Matrix: Startups/SMEs
3.7.1 Progressive Companies
3.7.2 Responsive Companies
3.7.3 Dynamic Companies
3.7.4 Starting Blocks
3.7.5 Competitive Benchmarking: Startups/SMEs
3.7.5.1 Detailed list of key startups/SMEs
3.7.5.2 Competitive benchmarking of key startups/SMEs
3.8 Competitive Scenario
3.8.1 Service Launches
3.8.2 Deals
3.8.3 Expansions
4.1 ProPharma Group Holdings, LLC
4.1.1 Business overview
4.1.2 Products/Solutions/Services offered
4.1.3 Recent developments
4.2 Caidya
4.2.1 Business overview
4.2.2 Products/Solutions/Services offered
4.2.3 Recent developments
4.3 CTI Clinical Trial & Consulting
4.3.1 Business overview
4.3.2 Products/Solutions/Services offered
4.3.3 Recent developments
4.4 Precision Medicine Group, LLC
4.4.1 Business overview
4.4.2 Products/Solutions/Services offered
4.4.3 Recent developments
4.5 Celerion
4.5.1 Business overview
4.5.2 Products/Solutions/Services offered
4.5.3 Recent developments
4.6 BioAgile Therapeutics Private Limited
4.6.1 Business overview
4.6.2 Products/Solutions/Services offered
4.6.3 Recent developments
4.7 Lotus Clinical Research, LLC
4.7.1 Business overview
4.7.2 Products/Solutions/Services offered
4.7.3 Recent developments
4.8 FGK Clinical Research GmbH
4.8.1 Business overview
4.8.2 Products/Solutions/Services offered
4.8.3 Recent developments
4.9 GreenLight Clinical
4.9.1 Business overview
4.9.2 Products/Solutions/Services offered
4.9.3 Recent developments
4.10 Navitas Life Sciences
4.10.1 Business overview
4.10.2 Products/Solutions/Services offered
4.10.3 Recent developments
4.11 Guires Inc.
4.11.1 Business overview
4.11.2 Products/Solutions/Services offered
4.11.3 Recent developments
4.12 Alira Health
4.12.1 Business overview
4.12.2 Products/Solutions/Services offered
4.12.3 Recent developments
4.13 Catalyst Clinical Research
4.13.1 Business overview
4.13.2 Products/Solutions/Services offered
4.13.3 Recent developments
4.14 Dove Quality Solutions
4.14.1 Business overview
4.14.2 Products/Solutions/Services offered
4.14.3 Recent developments
4.15 Firma Clinical Research
4.15.1 Business overview
4.15.2 Products/Solutions/Services offered
4.15.3 Recent developments